Introduction
Within the cattle industry, embryo loss
is a major concern. The cost to have an unsuccessful breeding session between
cows is an undue expense to ranchers and farmers. The first part of the
pregnancy is the period in which most embryo loss occurs. In this period, there
is a 10%-20% chance that a cow will lose her embryo (McSweeney). Often this
termination is due to a lack of maternal recognition. Making it so that the
mother recognizes the pregnancy could make it easier for the pregnancy to
advance to full term (White, et al., 2011).
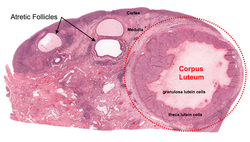
A critical part of mammalian pregnancy is the corpus luteum (CL). Within a female mammal, the corpus luteum is responsible for the initial production of progesterone that plays an essential part in maintaining a pregnancy in its beginning stages (Shams & Berisha, 2004). In the pregnant cow, the embryo trophoblastic cells secrete a cytokine named interferon-Tau. When interferon-Tau reaches strong concentrations, it triggers a response to the mother indicating maternal recognition of pregnancy.
The corpus luteum is a temporary hormone releasing endocrine gland that produces progesterone in stereoidogenic cells named luteal cells (Nancarrow, et al., 1973). It releases the steroid hormone progesterone regardless if the cow is pregnant or not. If the cow is pregnant, the CL will secrete progesterone for part of the pregnancy until the cow’s placenta takes over the production of progesterone (Shams & Berisha, 2004). Progesterone is responsible for the female cow's estrus cycle and is partially responsible for maintaining and establishing a pregnancy (Corpus Luteum , 2014 ). However, if the cow is not pregnant the CL needs to be regressed so that the estrus cycle may begin again.
In the non-pregnant cow, PGF2α is synthesized in the uterus and is known as a luteolytic hormone which initiates an apoptotic cell signaling cascade. Prostaglandin then flows to the ovary and regresses the CL in what is also known as luteolysis (Fields & Fields, 1996). Without the release of critical levels of interferon-Tau, PGF2α causes significant reduction of CL progesterone synthesis and the reduction of actual luteal tissue and the termination of the pregnancy (Nancarrow, et al., 1973).
The addition of fish oil containing omega-3 fatty acids has been suggested to play a role in maintaining a pregnancy in bovine by creating a shift in proteins found in lipid microdomains which might affect PGF2α synthesis (White, et al., 2011). Lipid microdomains are small ordered regions of the plasma membrane comprised of cholesterol and sphingolipids involved in signal transduction. It is postulated that lipid microdomains act as a dock for the PGF2α hormone and its FP receptor, coupling this pair to a G protein cell signaling complex. The dispersion of lipid microdomains could disrupt PGF2α and its bound FP receptor and inhibit the apoptotic PGF2α cell signaling complexes so that pregnancy has more time to reach full term (Pike, 2008). This is perhaps due to the separation of lipid microdomains, cholesterol, sphingolipids, and other associated proteins caused by the incorporation of the omega-3 fatty acids and their polyunsaturated long chain fatty acid tails into the plasma membrane of the CL (White, et al., 2011).
The stimulation of the uterus produces PGF2α (White, et al., 2011). It will act on the corpus luteum, significantly reducing progesterone synthesis (functional regression) and regressing the luteal tissues (structural regression) (Shams & Berisha, 2004). If a cow is pregnant, the embryo must control the uterine and possibly luteal PGF2α metabolism in an effort to maintain the pregnancy. A bovine embryo controls PGF2α by releasing interferon-Tau until it reaches critical levels. If the embryo does not control PGF2α, the pregnancy will be terminated in order to re-initiate the estrus cycle (White, et al., 2011). This termination is caused by a lack of critical levels of interferon-Tau released by the embryo which in turn tells the uterine to release PGF2α and regress the CL. It is possible that PGF2α might be able to be controlled through the addition of fish oil which contains omega-3 fatty acids (White, et al., 2011). This would occur through the lack of PGF2α binding to the FP receptors found within the cell.
A critical part of mammalian pregnancy is the corpus luteum (CL). Within a female mammal, the corpus luteum is responsible for the initial production of progesterone that plays an essential part in maintaining a pregnancy in its beginning stages (Shams & Berisha, 2004). In the pregnant cow, the embryo trophoblastic cells secrete a cytokine named interferon-Tau. When interferon-Tau reaches strong concentrations, it triggers a response to the mother indicating maternal recognition of pregnancy.
The corpus luteum is a temporary hormone releasing endocrine gland that produces progesterone in stereoidogenic cells named luteal cells (Nancarrow, et al., 1973). It releases the steroid hormone progesterone regardless if the cow is pregnant or not. If the cow is pregnant, the CL will secrete progesterone for part of the pregnancy until the cow’s placenta takes over the production of progesterone (Shams & Berisha, 2004). Progesterone is responsible for the female cow's estrus cycle and is partially responsible for maintaining and establishing a pregnancy (Corpus Luteum , 2014 ). However, if the cow is not pregnant the CL needs to be regressed so that the estrus cycle may begin again.
In the non-pregnant cow, PGF2α is synthesized in the uterus and is known as a luteolytic hormone which initiates an apoptotic cell signaling cascade. Prostaglandin then flows to the ovary and regresses the CL in what is also known as luteolysis (Fields & Fields, 1996). Without the release of critical levels of interferon-Tau, PGF2α causes significant reduction of CL progesterone synthesis and the reduction of actual luteal tissue and the termination of the pregnancy (Nancarrow, et al., 1973).
The addition of fish oil containing omega-3 fatty acids has been suggested to play a role in maintaining a pregnancy in bovine by creating a shift in proteins found in lipid microdomains which might affect PGF2α synthesis (White, et al., 2011). Lipid microdomains are small ordered regions of the plasma membrane comprised of cholesterol and sphingolipids involved in signal transduction. It is postulated that lipid microdomains act as a dock for the PGF2α hormone and its FP receptor, coupling this pair to a G protein cell signaling complex. The dispersion of lipid microdomains could disrupt PGF2α and its bound FP receptor and inhibit the apoptotic PGF2α cell signaling complexes so that pregnancy has more time to reach full term (Pike, 2008). This is perhaps due to the separation of lipid microdomains, cholesterol, sphingolipids, and other associated proteins caused by the incorporation of the omega-3 fatty acids and their polyunsaturated long chain fatty acid tails into the plasma membrane of the CL (White, et al., 2011).
The stimulation of the uterus produces PGF2α (White, et al., 2011). It will act on the corpus luteum, significantly reducing progesterone synthesis (functional regression) and regressing the luteal tissues (structural regression) (Shams & Berisha, 2004). If a cow is pregnant, the embryo must control the uterine and possibly luteal PGF2α metabolism in an effort to maintain the pregnancy. A bovine embryo controls PGF2α by releasing interferon-Tau until it reaches critical levels. If the embryo does not control PGF2α, the pregnancy will be terminated in order to re-initiate the estrus cycle (White, et al., 2011). This termination is caused by a lack of critical levels of interferon-Tau released by the embryo which in turn tells the uterine to release PGF2α and regress the CL. It is possible that PGF2α might be able to be controlled through the addition of fish oil which contains omega-3 fatty acids (White, et al., 2011). This would occur through the lack of PGF2α binding to the FP receptors found within the cell.
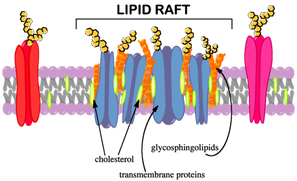
Omega-3 fatty acids are classified as essential polyunsaturated fatty acids. Research suggests that the addition of fish oil or fish meal containing omega-3 fatty acids to a cow’s diet could decrease the cell signaling response to PGF2α (White, et al., 2011). Lipid microdomains contain high concentrations of cholesterol and sphingolipids and are between 5-500 nanometers in size (Pike, 2003). Multiple proteins are found in lipid microdomains. Many of these proteins are associated with cell signaling (Pike, 2008). Because of this, it is believed that lipid microdomains might be connected to the signal regulation within a cell (Pike, 2003). Two proteins that make up the greatest amount of lipid microdomains are caveolin and flotillin. Both types of proteins are believed to play a role in cell transduction (Pike, 2003). Since lipid microdomains are connected to cell signaling, it is believed that PGF2α docks onto receptors found within these lipid microdomains and causes the signals that regress the CL and terminates the pregnancy (Nancarrow, et al., 1973). If these lipid microdomains are disrupted so that PGF2α receptors could not dock and transmit signals into the cell, this might inhibit regression of the CL and allow the maternal recognition of pregnancy and give the pregnant cow a chance at full term gestation (White, et al., 2011).
The disruption of caveolin and flotillin which are associated with lipid microdomains causes a shift from sucrose fractions 1-4 to the bulk lipids, fractions 5-10 (Calder & Yaqoob, 2006). The fish oil containing omega-3 fatty acids could affect the cholesterol and sphingolipid levels within the lipid microdomains which would decrease cell signaling due to a change in the protein arrangement within the cell (White, et al., 2011). Therefore if omega-3 fatty acids are incorporated into plasma membranes and disrupt lipid microdomains, a shift of flotillin and caveolin from fractions 1-4 to fractions 5-10 would be seen. Thusly, we hypothesize that the incorporation of omega-3 fatty acids found in fish oil to bovine luteal cells will cause a shift in proteins associated with lipid microdomains to bulk lipids indicating dispersion of the lipid microdomains. Due to this the purpose of this experiment was to determine the effect of fish oil containing omega-3 fatty acids on distribution of proteins flotillin and caveolin associated with lipid microdomains in bovine luteal cells.
The disruption of caveolin and flotillin which are associated with lipid microdomains causes a shift from sucrose fractions 1-4 to the bulk lipids, fractions 5-10 (Calder & Yaqoob, 2006). The fish oil containing omega-3 fatty acids could affect the cholesterol and sphingolipid levels within the lipid microdomains which would decrease cell signaling due to a change in the protein arrangement within the cell (White, et al., 2011). Therefore if omega-3 fatty acids are incorporated into plasma membranes and disrupt lipid microdomains, a shift of flotillin and caveolin from fractions 1-4 to fractions 5-10 would be seen. Thusly, we hypothesize that the incorporation of omega-3 fatty acids found in fish oil to bovine luteal cells will cause a shift in proteins associated with lipid microdomains to bulk lipids indicating dispersion of the lipid microdomains. Due to this the purpose of this experiment was to determine the effect of fish oil containing omega-3 fatty acids on distribution of proteins flotillin and caveolin associated with lipid microdomains in bovine luteal cells.
